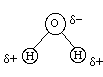
Water is a very unusual substance in that the density of the solid is less than the density of the liquid. If the densities were reversed water would freeze from the bottom up and there would be no life as we know it on earth. With the density of water being less than the density of the liquid, there is also a mixing of the lakes as the temperature drops (called a turnover). Water is a non-linear molecule and the oxygen atom draws electrons from the molecule toward itself (electronegativity difference -- will discuss later) so that the oxygen atom as a net negative charge and the hydrogen atoms have a net positive charge. These charges are not full (integer) charges and are represented by the small Greek letter delta -- there is no html code for Greek letters (except mu) so I will use "d" for delta. The water structure is

with an H-O-H angle of about 105 degrees. Due to the charges on the hydrogen atoms and the oxygen atom, there is a charge separation for the molecule (negative on one end and positive on the other end). Thus this molecule possesses a dipole moment (a dipole is a species with a charge separation like
q+ -------- q- <-----d----->and the dipole moment (D) is D = q*d (where q is the charge and d is the distance between the charge centers). A molecule with a permanent dipole like this is called a polar molecule. Due to its polar nature, the water molecule can attach itself to other water molecules by what is called a hydrogen bond (to be discussed later):
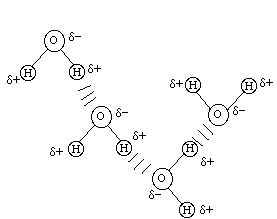
The polar nature of the water molecule allows it to attach itself to other molecules which have charges leading to rather stable solutions (called hydration). Methanol has the structure:
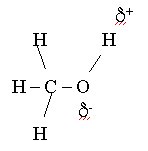
and water and methanol will attach themselves to each other via a hydrogen bond also. Ionic compounds like NaCl also have positive and negative "ends" and thus the positive hydrogen atoms from water will attach to the negative Cl anion and the negative oxygen atom from water will attach itself to the positive Na cation and this process will dissolve the NaCl ionic matrix (remember no such thing as a single NaCl molecule).
An electrolyte is a compound which dissociates in a water solution into its component ions like
NaCl ---> Na+ + Cl-
They are called electrolytes because pure water does not conduct electricity, but when an ionic compound is dissolved in water the solution will conduct electricity. We further categorize electrolytes into strong electrolytes and weak electrolytes.
Strong electrolytes(generally ionic compounds) are almost totally dissociated into their component ions and thus form a solution which is a very good electrical conducting solution.
AB ---> A+ + B- These are strong acids (hydrochloric, sulfuric, nitric), strong bases (NaOH, KOH), and soluble salts.
Weak electrolytes(generally molecular compounds) are not totally dissociated into their component ions and thus form a solution which is a weak electrical conducting solution.
AB --> A+ + B- <--These are weak acids (acetic acid), weak bases (ammonia), and poorly soluble salts.
If a molecule is dissolved in water and there is no conduction of electricity, the molecule is called a nonelectrolyte. Examples of a nonelectrolyte are sucrose (C12H22O11 and methanol -- structure above).
![]() Go to this site for a picture of ion hydration if you have a fast connection.
Go to this site for a picture of ion hydration if you have a fast connection.
| Now take a practice quiz to help you understand if you understand the basic concepts. |
| You must use your real name when it asks for a name. |
| The test will only submit when you have answers all of the questions correctly. |
| If you are not taking this course for credit please do not answer all the questions correctly for I don't want to be flooded with email answers to the tests. |

Web Author: Dr. Leon L. Combs
Copyright ©1999 by Dr. Leon L. Combs - ALL RIGHTS RESERVED